First we continued to go over yesterday's homework.
EC said that when you pump a basketball up, its not really
full because the particles inside are all moving around
GS continued her thought saying that air has a lot of empty space in it.
To test whether it is full or not, we could try to pump more air into the second picture.
Then we drew these two pictures. The first is before we pump more air in, and the second is after we pump more air in.
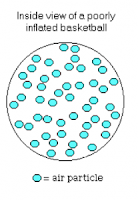
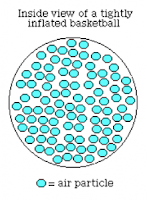
The dots represent particles. There are more particles in the second than the first. This diagram does not include arrows to show direction and speed, but the particles in the first go much fatser
DB asked a good question. "Why, if the particles are moving, doesn't the ball change shape?"
That brought us to the balloon question from last night. As we pump air into the balloon more particles go in, move around, and push the sides of the balloon out.
SA and Finley each pushed on a piece of paper. SA represented the air pressing against the outside of the balloon and Finley represented the air pressing on the inside. The paper didn't move so the balloon doesn't expand. Then Finley stopped pushing so SA moved the paper forward. This shows that the balloon would expand.
When you are blowing it up the push on the inside is stronger than the push on the outside. That is why it expands. When you stop blowing the force on the inside and outside are equal. That is called an equilibrium. When you put it in a vacuum, the force outside is less than than the force inside again.
Then we went over the cup question. Finley held up a cup and asked if it was full.
MA said it was full of air.
To test this we would turn it upside down and slowly push the cup into water. We put a paper boat on the water and pushed it down.
We did this test in class. The boat did not get wet once we brought the cup back up. A tiny bit of water was in the cup
In short the water did not go into the glass because the air was pushing again the water and the water was pushing against the air.
The reason a little bit of water did was because air is partially empty space. The water was trying to compress the air so it could get in. That is why there is less empty space at the end and a little bit of water got in.
This is like an old scuba diver helmet that contains air and water can't get it.
It is also the same concept with bubbles. The particles of air are very compressed so the bubble has a lot of force pushing outward.
Then we started talking about conservation. Its like energy. 10J K + 10 J Ug always equals 20 J.
Volume is not conserved because when you add 10 ml of some particle that can get absorbed by water to 10 ml of water. You will NOT get 20 ml of stuff.
When something can's go into the water it still will not be conserved. The water goes into the other material.
Its like sand. The water goes into the sand. Like at the beach, the sand by the water is wet.
Then we tested whether mass can be conserved.
Finley poured 306.2 grams of water into a graduated cylinder. The cylinder itself weighed 52.2. We have five grams of sugar. What would happen? Will we get 311.2 grams? We checked. It was 311.1. Because of uncertainty mass is conserved.
This weekends home work is... NO HOMEWORK!
I have a question. If you put a solid in water with no empty space, would the volume be conserved? Finley said volume is never conserved, but in this case, where would the water go?
MK (post 3)
I tried to put colors up, but the computer wouldn't let me
ReplyDeleteMK
I am going to try to answer your question.
ReplyDeleteI think if you put a solid in water with no empty space, the container would overflow. If the container would not allow the water to overflow, the block would not be able to be pushed in. So the volume wouldn't be conserved. Hope this helps!
EC